The number of research and development programmes is increasing year by year with a strong focus in three key areas: respiratory, neonatology and rare disease & special care.
Respiratory is a core area of R&D expertise, which has delivered significant value for the company and continues to expand to meet areas of unmet medical need in asthma, Chronic Obstructive Pulmonary Disease (COPD) and other respiratory diseases
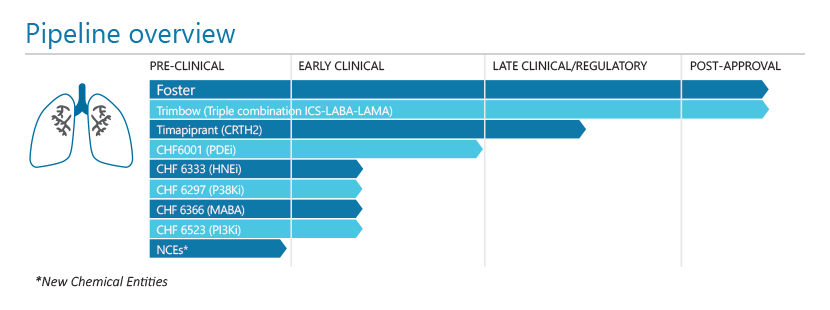
Inuvair is a key revenue driver for the future of the company, but it also remains a highly significant element of R&D activity as we roll out a powerful life cycle management program.
There is a substantial commitment to a very attractive therapeutic opportunity with our Triple programme. Our product candidate, now approved in Europe, contains three active ingredients in a single inhaler that will ensure effective anti-inflammatory therapy combined with maximum bronchodilation throughout the entire respiratory tree.
The exciting pipeline of new molecules continues to show significant promise. These programmes include novel anti-inflammatory and bronchodilator molecules, and also innovative “double” active molecules addressing two different pharmacological targets.
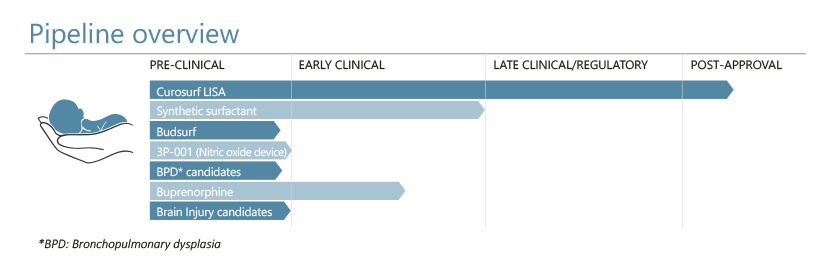
Through the clinical success of Curosurf in treating neonatal respiratory distress syndrome, Chiesi has established an impressive platform in neonatology. In this area of high unmet medical need R&D programmes include novel and less-invasive methods for the targeted delivery of Curosurf to the lung of the infant. The proprietary synthetic surfactant is undergoing clinical trials with emerging data supporting the safety, efficacy and clinical utility of this potential new addition to surfactant replacement therapy options.
As we expand our pipeline behind these ground-breaking respiratory-focused products, new programmes have been developed and added for candidates for the treatment of other neonatal conditions. For example, we are advancing a program for a novel therapeutic for the treatment of Neonatal Opioid Withdrawal Syndrome (NOWS). Moreover, there are currently no effective approved pharmacological therapeutics for neonatal brain injury: a relatively common condition associated with significant levels of morbidity and mortality in newborns.
Chiesi’s R&D model in the special care area includes some significant partnerships with selected companies skilled in the study of medicines for rare diseases. This enables the company to access advanced therapeutic platforms and promising new therapies in newer areas of the pipeline
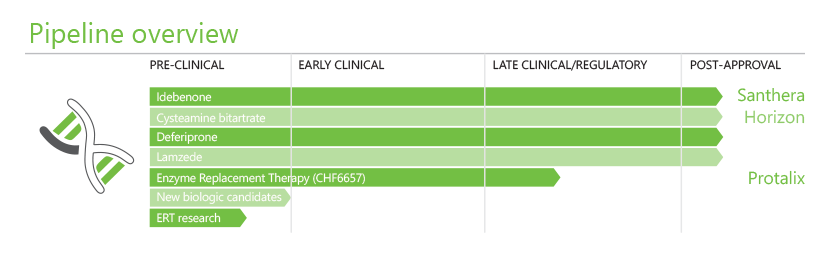
The acquisition and now full integration of Zymenex, the Scandinavian-based biotechnology company, represented a bold step to bring into Chiesi key product candidates and competencies in the biotech area and design therapies for treating extremely rare diseases through enzyme replacement therapy. The most advanced candidate, Lamzede, is now approved in Europe.
In partnership with Veloxis we have gained approval for a once-daily formulation of the immunosuppressant tacrolimus for the prophylaxis of solid organ transplant rejection.